Mechanics of cancer metastasis
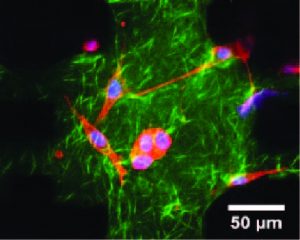
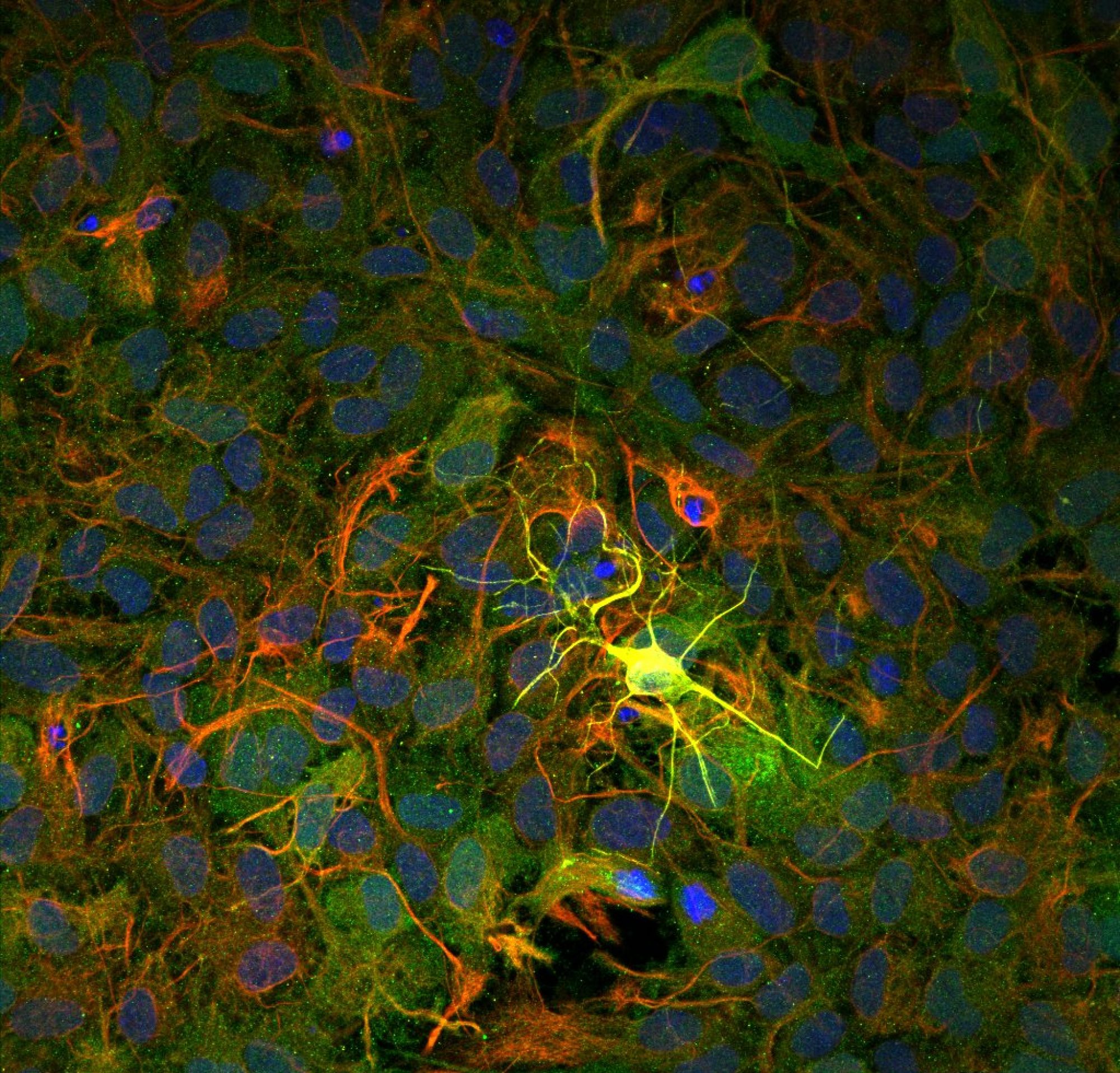
Cancer deaths are mostly caused by metastasis when cancer cells migrate away from the primary tumor to invade other organs. Tumor growth and metastatic transition of cancer cells are determined by a dynamic crosstalk between the malignant cells and the tumor microenvironment. The latter consists of stromal cells, mostly dominated by cancer associated fibroblasts (CAF) and extracellular matrix (ECM). CAFs generate mechanical contractile force. They remodel the matrix by mechanically pulling and aligning the ECM fibers, and by releasing ECM crosslinkers. Remodeled matrix favors migration of invasive cancer cells. In spite of known contractility, the mechanistic role of CAF force on cancer progression remains elusive. While the current literature views the many participants of tumor microenvironment as interacting emergent entities, it does not identify any potential universal driver that mediates this emergence. And yet, if such a driver can be identified and characterized, then its function can be interrupted to disrupt the circuit of cancer progression. We argue, based on our preliminary evidence, that CAF force can be such a driver. We are currently exploring this FORCE (FORce control of Cancer tumor µEnvironment) hypothesis: CAFs promote metastatic progression through force-dependent release of growth factors for cancer cells, and ECM remodeling. We have developed a novel sensor (Lab Chip, 2019,19 (7), 1153-1161 https://doi.org/10.1039/C8LC01273C) to address these questions. It is now possible to form an in vitro cancer tumor integrated with the sensor (video above), which monitors cell forces, migration, remodeling of ECM in situ as a function of time (see image and video below, Science Advances 7 (15) (2021) eabf2629. https://doi.org/10.1126/sciadv.abf2629).